Cancer and heart disease vaccinations by end of decade 2023
Experts say a pioneering collection of new vaccinations for many diseases, including cancer, might save millions of lives. A major pharmaceutical company expects cancer, cardiovascular, autoimmune, and other vaccines to be ready by 2030.
Innovative vaccines, according to a pharmaceutical company, could save millions of lives.
These vaccines are also showing “tremendous potential,” with some experts stating the Covid shot has “unspooled” 15 years of advancement in 12 to 18 months.
Moderna’s chief medical officer, Dr. Paul Burton, believes the business can deliver such therapies for “all sorts of illness areas” in five years.
How personalized vaccines against cancer work
A top coronavirus vaccine maker is researching cancer vaccinations for distinct tumor types.
Burton stated, “We will have that vaccine and it will be very successful and save many hundreds of thousands, if not millions of lives. I think we can deliver global customized cancer vaccinations against numerous tumor types.”
He also suggested that a single injection might protect susceptible patients from Covid, flu, and respiratory syncytial virus (RSV), that mRNA treatments could treat unusual disorders without medications. mRNA therapies educate cells to generate a protein that activates the immune system.
Burton said, “I think we will have mRNA-based therapies for rare diseases that were previously undruggable, and I think that 10 years from now, we will be approaching a world where you truly can identify the genetic cause of a disease and, with relative simplicity, go and edit that out and repair it using mRNA-based technology.”
Scientists warn that if funding isn’t sustained, the last three years’ “order of magnitude” gains would be squandered.
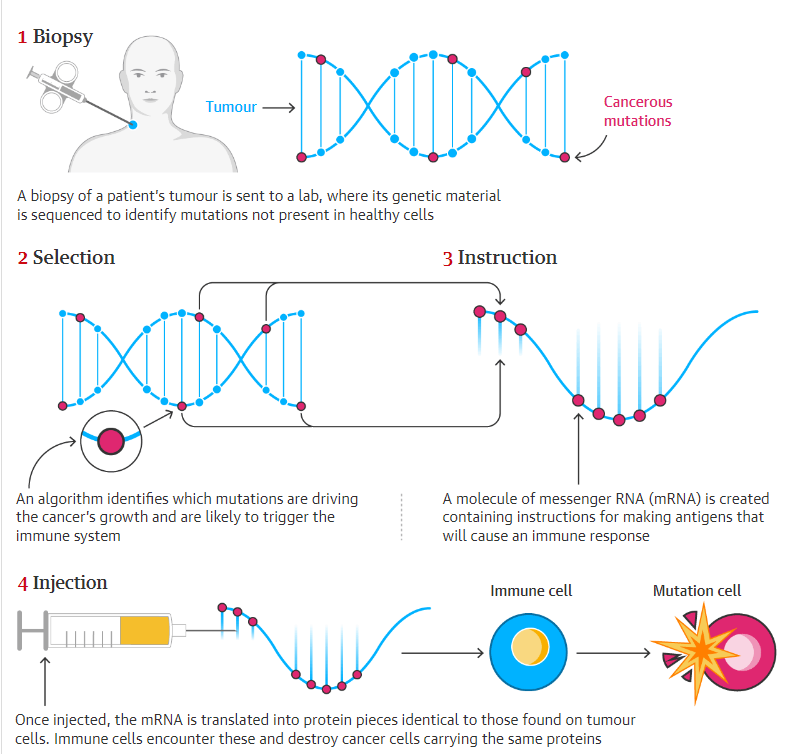
mRNA directs protein production. Cells can release proteins for our immune system by injecting a synthetic form. An mRNA-based cancer vaccine would notify the immune system to a developing cancer so it may attack and eliminate it without harming healthy cells.
This entails finding protein fragments on the surface of cancer cells that are not present on healthy cells and most likely to activate an immune response, then generating mRNA to guide the body to create them.
Doctors biopsy a patient’s tumor and send it to a lab to sequence its genetic material for alterations.
A machine learning system determines which mutations cause cancer growth. It also learns which regions of these mutant proteins are most likely to cause an immunological response. Next, mRNAs for the best antigens are made and packaged as a personalized vaccination.
Burton added, “I believe what we have learnt in recent months is that if you ever thought that mRNA was only for infectious illnesses, or just for Covid, the evidence today is that that’s definitely not the case.
We treat cancer, infectious illness, cardiovascular disease, autoimmune disease, and uncommon disease. All those research have showed great promise.”
Moderna’s experimental RSV mRNA vaccine was 83.7% efficient at preventing at least two symptoms, such as cough and fever, in persons 60 and older in a late-stage study in January. Based on these evidence, the US Food and Drug Administration (FDA) gave vaccination breakthrough treatment status, expediting regulatory consideration.
Based on recent melanoma patient findings, the FDA gave Moderna’s personalized cancer vaccine the same classification in February.
Burton added, “I think the pandemic sped [this technology] up by an order of magnitude. It has also helped us to scale up manufacturing, so we’re really adept at creating vast volumes of vaccine quickly.”
Pfizer and BioNTech are also recruiting for a late-stage clinical study of an mRNA-based influenza vaccine and other infectious illnesses, including shingles. “The learnings from the Covid-19 vaccine development process have shaped our general approach to mRNA research and development, and how Pfizer performs R&D more broadly,” stated a Pfizer spokeswoman. We learned a decade of science in one year.”
Novavax’s Covid jab has benefited from the outbreak. The jab fools the immune system into attacking a germ.
Novavax president of research and development Dr. Filip Dubovsky said: “There has been a significant acceleration, not just of standard vaccine technologies, but also unique ones that hadn’t previously been carried through licensure. mRNA and our vaccine are examples.
Dr. Richard Hackett, CEO of Cepi, said the pandemic has shortened development times for several unvalidated vaccine platforms. “It meant that things that may have unspooled over the following decade or perhaps 15 years, were compacted down within a year or a year and a half,” he said.
Prof. Andrew Pollard, head of the Oxford Vaccine Group and chair of the UK Joint Committee on Vaccination and Immunisation (JCVI), stated, “There’s no doubt there’s a lot more interest in vaccinations. What happens next?
With the possibility of larger conflict in Europe, this concentration on vaccinations may be lost, wasting the momentum and scientific advances acquired during the epidemic. Pollard thinks this is wrong.
“If you take a step back to look about what we are prepared to invest on during peacetime, like maintaining a significant military for most countries… Since pandemics are inevitable, they are as dangerous as military threats. Yet we’re not investing even the cost of one nuclear submarine.”
